 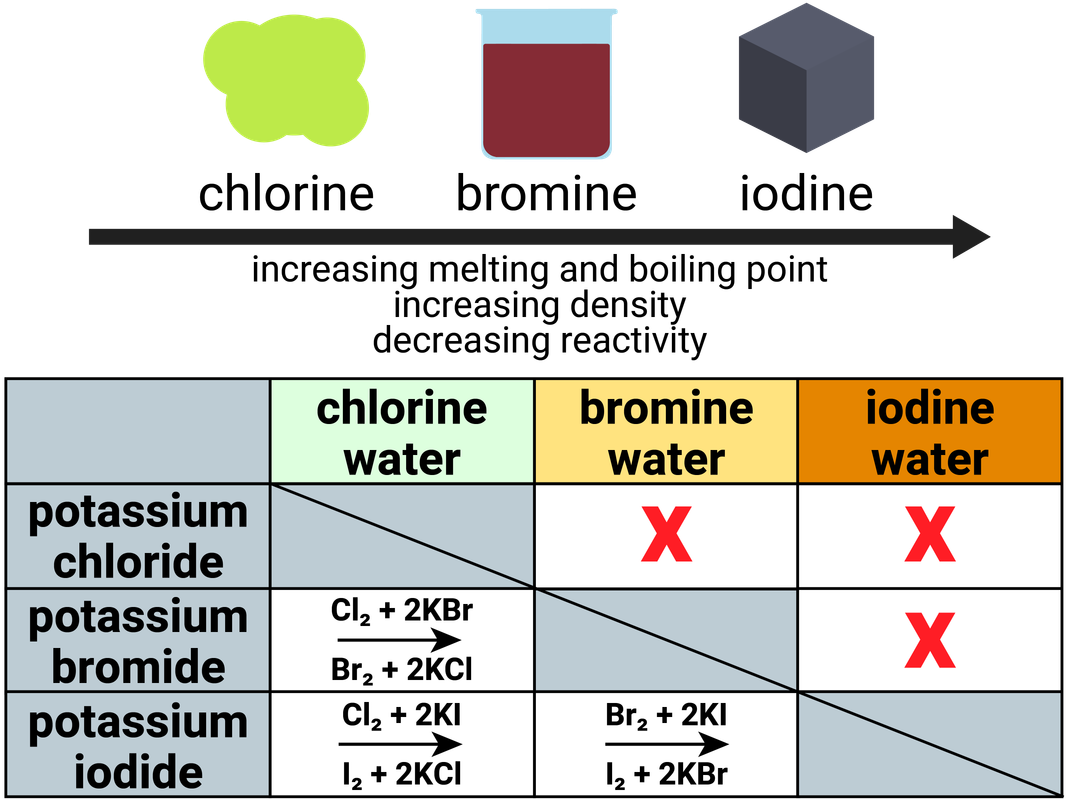
When an electron is added to such a small atom, increased electron–electron repulsions tend to destabilize the anion. The reaction was observed, and the temperature of the solutions was taken before and after the metal was placed in the solution, as well as a Litmus test. First ionisation energy increases across the period. Period 3 shows trends in atomic properties: Ionic radius decreases across the period. 
It contains the elements sodium, magnesium, aluminium, silicon, phosphorus, sulphur, chlorine, and argon. Step 1: Identify the elements present and their location on the periodic table. Results and Discussion: The periodic table is arranged in a way where the properties of elements are recognized by placement in the table. Period 3 is the third row in the periodic table. In contrast to the chemistry of the second-period elements, the chemistry of the third-period elements is more representative of the chemistry of the respective group.ĭue to their small radii, second-period elements have electron affinities that are less negative than would be predicted from general periodic trends. Steps for Explaining the Periodic Trends in Reactivity. The anomalous chemistry of second-period elements results from three important characteristics: small radii, energetically unavailable d orbitals, and a tendency to form pi (π) bonds with other atoms. Visualize trends, 3D orbitals, isotopes, and mix compounds. Consequently, the elements of the third period (n = 3: Na, Mg, Al, Si, P, S, and Cl) are generally more representative of the group to which they belong. Interactive periodic table showing names, electrons, and oxidation states. The chemistry of the second-period element of each group (n = 2: Li, Be, B, C, N, O, and F) differs in many important respects from that of the heavier members, or congeners, of the group. 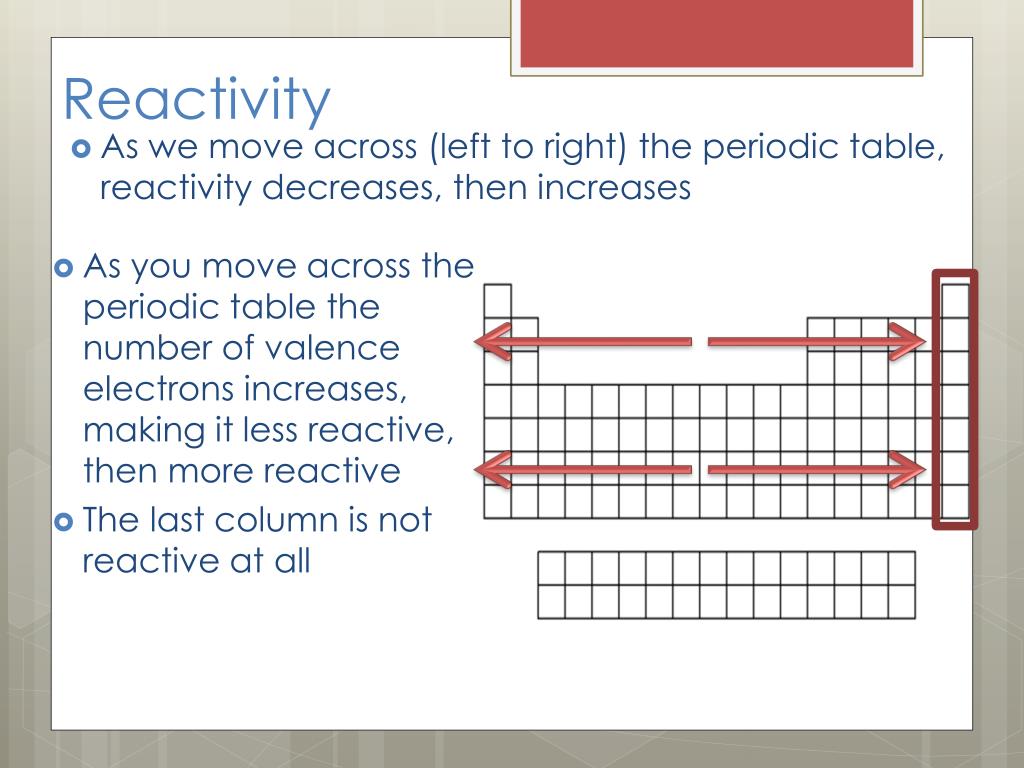
A common characteristic of most Alkali Metals is their ability to displace H 2 (g) from water. Use this chart to see at a glance the periodic table trends of electronegativity, ionization energy, atomic radius, metallic character, and electron affinity. Unique Chemistry of the Lightest Elements Many of these chemical reactions behave in trends that can be categorized using the periodic table. The semimetals lie along the diagonal line separating the metals from the nonmetals and exhibit intermediate properties. Consequently, the elements in the upper right of the periodic table are the smallest and most electronegative the elements in the bottom left are the largest and least electronegative. Page notifications Off Share Table of contents Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element. In contrast, atomic size decreases from left to right and from bottom to top. Ionization energies, the magnitude of electron affinities, and electronegativities generally increase from left to right and from bottom to top. \): Summary of Periodic Trends in Atomic Properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
