 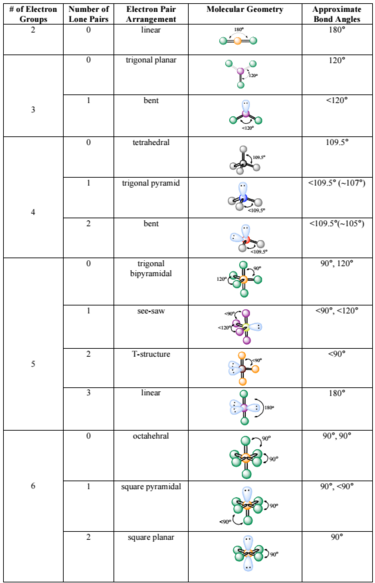 
Once again, we have a compound that is an exception to the octet rule. Each fluorine has three lone pairs, Bromine has two lone pairs. Three fluorines are bonded to a central bromine. The hydronium ion, H 3 O +, forms when acids are dissolved in water. The bromine atom has seven valence electrons, and each fluorine has seven valence electrons, so the Lewis electron structure is. According to the VSEPR theory, if the H2S molecule ion has an AX2N2 generic formula, the molecular geometry and electron geometry will both be tetrahedral or V-bent-shaped forms. (b) Two of the electron regions are lone pairs, so the molecular structure is bent. The general molecular geometry formula for H2S is AX2N2. With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position. 9: (a) H2O has four regions of electron density around the central atom, so it has a tetrahedral electron-pair geometry. If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. Vsepr molecular geometry chart Vsepr and molecular geometries reference table by mj Molecular structure by. However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. Electron group geometries refer to the five geometries: linear, trigonal planar, tetrahedral, trigonal bipyramidal, or octahedral. 
We designate SF 4 as AX 4E it has a total of five electron pairs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
